Previous clinical trials for filgotinib for RA
Phase 2a proof-of-concept trial
In November 2011, Galapagos announced topline data from a Phase 2a proof-of-concept trial (GLPG0634-CL-201), a four-week trial performed in RA patients with insufficient response to MTX. This trial was a randomized, double-blind, placebo-controlled trial that was conducted in a single center. A total of 36 patients were randomized in a 1:1:1 allocation ratio to receive filgotinib 100 mg (twice-daily), 200 mg (daily) or placebo, respectively, while continuing their stable dose regimen of MTX. All randomized patients completed the trial.
In the trial, ACR20 at week 4 was achieved by approximately 92% (p-value versus placebo = 0.0094), 75% (p-value versus placebo = 0.0995), and 33% in the 100 mg (twice-daily), 200 mg (daily) and placebo groups, respectively, and up to 40% of the filgotinib-treated patients went into either disease remission or low disease activity. The difference in number of ACR20 responders at week 4 was statistically significant for the pooled GLPG0634 group versus the placebo group (p-value versus placebo = 0.0067).
No serious adverse events, or SAEs, were reported on patients who received active treatment with various doses and dose regimens of filgotinib and there were also no permanent discontinuations among patients treated with filgotinib. Median laboratory values and p-values were visually inspected for trends over time, however, no statistical analysis on trends over time was performed. No clinically relevant trends or changes were apparent from these analyses, except for a decrease in platelet count in both filgotinib treatment groups. Vital signs and electrocardiogram, or ECG, parameters were not influenced by filgotinib. Overall, the results of this proof-of-concept trial in patients with RA demonstrated that a daily dose of 200 mg of filgotinib on top of MTX shows promising activity and was generally well-tolerated over four weeks of treatment.
Phase 2a dose-ranging trial
In November 2012, Galapagos announced topline data from a follow-up Phase 2a dose-ranging trial (GLP0634- CL-202) to confirm the safety profile observed in the Phase 2a proof-of-concept trial. This trial was a four-week, randomized, double-blind, placebo-controlled, dose-ranging trial performed in patients with active RA who had an inadequate response to MTX and was conducted in four countries and involved 19 centers. A total of 91 patients were randomized in a 1:1:1:1:1 allocation ratio to receive once-daily regimens of 30 mg filgotinib, 75 mg filgotinib, 150 mg filgotinib, 300 mg filgotinib or placebo during four weeks, respectively.
In this trial, ACR20 by week 4 was achieved by 35% (p-value versus placebo = 0.736), 55% (p-value versus placebo = 0.456), 40% (p-value versus placebo = 0.834), 65% (p-value versus placebo = 0.111), and 41% for doses 30 mg, 75 mg, 150 mg, 300 mg and placebo, respectively. Overall activity of filgotinib was confirmed across a wide panel of parameters. Some imbalances among treatment groups in demographic and disease characteristics, as well as the limited size of each treatment group, may explain the relatively high placebo ACR20 response rate and the apparently low ACR20 response rate of the 150 mg/day filgotinib dose group. Overall, more consistent and dose-related results across treatment groups were observed for objective measures of disease activity, such as serum C-reactive protein, and for assessments of disesase such as swollen joint counts (SJC), tender joint counts (TJC), compared with the subjects' subjective assessments, i.e. global disease and pain assessment or Health Associated Questionnaire – Disability Index (HAQ-DI). This was particularly evident in the 150mg dose group, in which subjects had a higher SJC and TJC at baseline than the other arms, and may have resulted in less perceived improvement in pain and global visual analog scale, or VAS, leading to a poor ACR response. We selected the 50, 100, and 200 mg doses for the DARWIN Phase 2b program based on the outcome of this trial.
No SAEs were reported on patients who received active treatment with various doses of filgotinib and there were also no permanent discontinuations among patients treated with filgotinib. No medically significant shifts from baseline in laboratory parameters evaluated were seen. Filgotinib was well-tolerated at all dosages. The safety profile in this trial was not different to the previous trials conducted on filgotinib. Vital signs and ECG parameters were not significantly influenced by filgotinib.
Phase 1
Galapagos evaluated filgotinib in healthy human volunteers in Phase 1 trials and did not achieve a maximum tolerated dose, even at a dose of 450 mg. Through its compound specific metabolic transformation, filgotinib has a one-day half-life, which may contribute to its once-daily, or QD, efficacy.
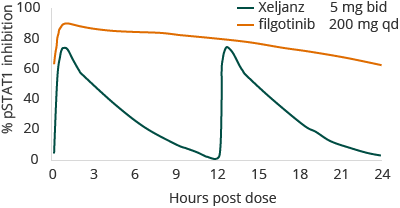
Furthermore, the potential for drug-drug interactions for filgotinib and its major metabolite was investigated in vitro, and confirmed with midazolam (marker for CYP3A4) in healthy volunteers and with MTX in patients. As filgotinib does not interact with Cytochromes P450 Enzymes, or CYP, and does not inhibit key drug transporters, we expect that it can be used with these concomitant drugs without dose adjustment of filgotinib or these concomitant medications.
