Our differentiating approach
In 2022, we entered the field of oncology, CAR-T, and antibody-therapy research and development through the acquisitions of CellPoint and AboundBio. The transactions provide us with end-to-end capabilities in CAR-T therapy development and offer the potential for a paradigm shift in the space through the implementation of a breakthrough, decentralized point-of-care manufacturing model, and cutting-edge fully human antibody-based capabilities to design next-generation CAR-Ts and biologicals.
Point-of-care manufacturing
Despite continued progress with current CAR-T cancer therapies, long lead times, costly central manufacturing and complex logistics continue to be limiting factors for large-scale capacity and broad patient access.
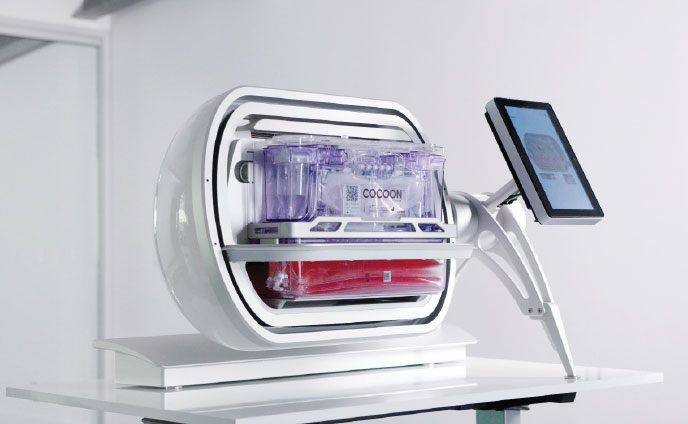
To address important limitations of current CAR-T treatments, CellPoint (a Galapagos company) has developed, in a strategic collaboration with Lonza, a novel decentralized delivery model designed to manufacture non-frozen CAR-T therapies at the point-of-care.
Through decentralized manufacturing, complex logistics and cryopreservation of the cells can be avoided, and the average vein-to-vein time can be drastically reduced from up to months for currently approved CAR-T therapies to 7 days with our CAR-T candidates currently observed in our clinical trials.
The proprietary platform consists of CellPoint’s end-to-end xCellit workflow management and monitoring software and Lonza’s Cocoon®, a functionally closed, automated manufacturing platform for cell therapies.
The novel point-of-care model is compliant with the EMA and FDA guidance for clinical trials.
Increase patient access with point-of-care manufacturing
Streamlining CAR-T Therapy
Antibody engineering capabilities
AboundBio (a Galapagos company) has developed several very large, diverse human antibody libraries in standard fragments of antigen binding (Fab), single-chain variable fragments (scFv), and unique variable (VH) domain formats. The team can rapidly (days to weeks) discover novel, high affinity, binders in multiple formats, engineer them if needed to improve their developability properties, and convert them for multiple uses including multi-specific, CARs, fusion proteins and antibody drug conjugates (ADCs). The proprietary methodologies to build large fully-human antibody-based libraries offer the potential to increase binder diversity, affinity and specificity; coverage of potential antigens; screening capacity; and probability of identifying a lead therapeutic antibody candidate.
In the field of oncology, AboundBio provides unique research capabilities for next generation CAR-T therapies that have the potential to deliver deeper and durable clinical responses, as well as additional drug modalities beyond small molecules.
Our new generation of fully human, multi-specific CAR-T constructs have the potential to transform patient outcomes through potentially more effective and longer-lasting care options, even in the event of relapse after previous CAR-T-cell therapy. Together with the decentralized CAR-T point-of-care manufacturing model, we aim to broaden patient access and ultimately hope to change their lives.
